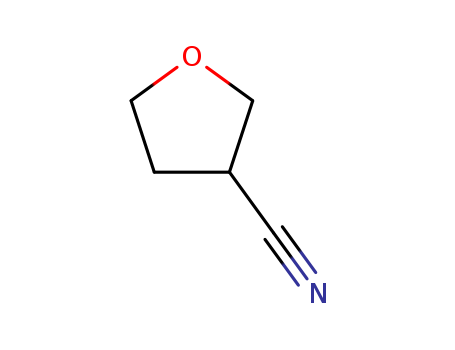
14631-44-8
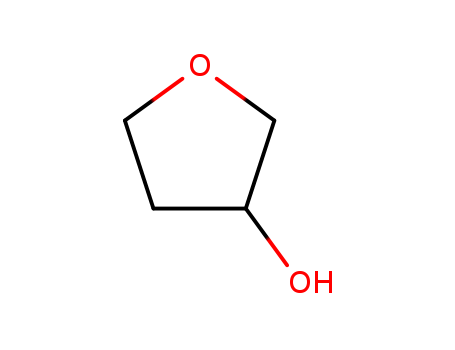
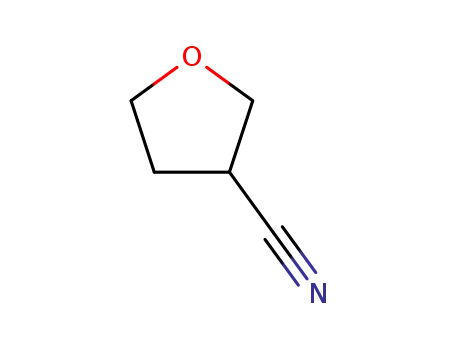
- Product Name:3-Cyanotetrahydrofuran
- Molecular Formula:C5H7NO
- Purity:99%
- Molecular Weight:97.1167
Product Details;
CasNo: 14631-44-8
Molecular Formula: C5H7NO
Manufacturer Sells Best Quality 3-Cyanotetrahydrofuran 14631-44-8 with stock
- Molecular Formula:C5H7NO
- Molecular Weight:97.1167
- Vapor Pressure:0.226mmHg at 25°C
- Refractive Index:1.445
- Boiling Point:207.41 °C at 760 mmHg
- Flash Point:84.339 °C
- PSA:33.02000
- Density:1.043 g/cm3
- LogP:0.54648
3-FURANCARBONITRILE, TETRAHYDRO-(Cas 14631-44-8) Usage
3-Cyano tetrahydrofuran is a chemical compound, specifically a cyclic ether with a nitrile functional group. The "tetrahydrofuran" part refers to the five-membered ring with one oxygen atom, and the "3-cyano" part indicates a nitrile group (-CN) is attached to the 3rd carbon of the ring. It's a useful intermediate in chemical synthesis, particularly in the creation of pharmaceuticals and other organic molecules.
InChI:InChI=1/C5H7NO/c6-3-5-1-2-7-4-5/h5H,1-2,4H2
14631-44-8 Relevant articles
Synthesis method for 3-aminomethyl tetrahydrofuran
-
Paragraph 0054; 0057; 0061; 0064; 0066; 0069; 0071; 0074, (2020/07/14)
The invention provides a synthetic metho...
Preparation method of 3-aminomethyltetrahydrofuran serving as dinotefuran intermediate
-
Paragraph 0044; 0046; 0049, (2018/07/07)
The invention relates to a preparation m...
Synthesis method of tetrahydrofuran-3-methylamine
-
Paragraph 0014; 0029, (2017/09/01)
The invention discloses a synthesis meth...
Preparation method for 3-aminomethyltetrahydrofuran
-
Paragraph 0013; 0022; 0023, (2017/02/24)
The invention discloses a preparation me...
14631-44-8 Process route
-
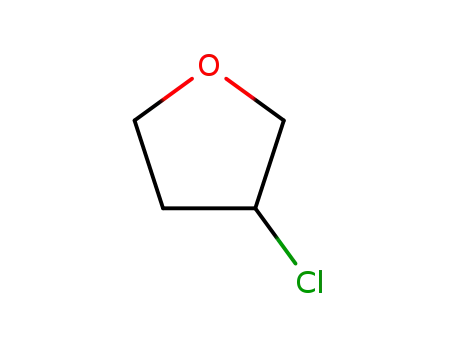
- 19311-38-7
3-chlorotetrahydrofuran

-
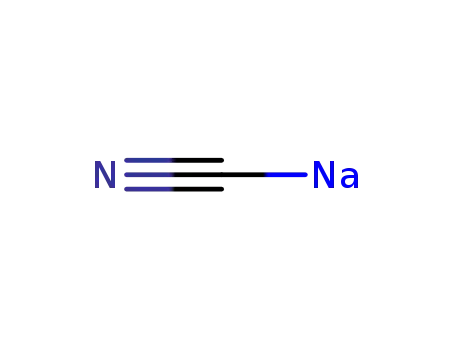
- 143-33-9,25596-52-5
sodium cyanide

-
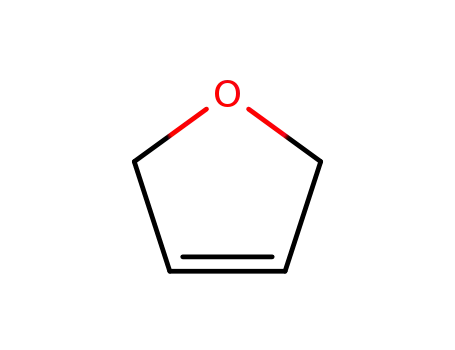
- 1708-29-8
2,5-dihydrofuran

-
- 14631-44-8
3-cyanotetrahydrofuran
| Conditions | Yield |
|---|---|
|
In dimethyl sulfoxide; at 130 ℃; for 4h; Product distribution / selectivity;
|
89.6 %Chromat. 8.8 %Chromat. |
|
In sulfolane; at 140 ℃; for 7h; Product distribution / selectivity;
|
55.6 %Chromat. 10.3 %Chromat. |
|
In 1,3-dimethyl-2-imidazolidinone; at 120 ℃; for 4h; Product distribution / selectivity;
|
59.6 %Chromat. 12.7 %Chromat. |
|
In 1-methyl-pyrrolidin-2-one; at 120 ℃; for 4h; Product distribution / selectivity;
|
54.3 %Chromat. 11.6 %Chromat. |
|
In DMF (N,N-dimethyl-formamide); at 150 ℃; for 5h; Product distribution / selectivity;
|
87.1 %Chromat. 10.1 %Chromat. |
|
In ethanol; at 140 ℃; for 7h; Product distribution / selectivity;
|
22.8 %Chromat. 15.3 %Chromat. |
|
In N,N-dimethyl acetamide; at 130 ℃; for 3h; Product distribution / selectivity;
|
52.7 %Chromat. 8.4 %Chromat. |
|
at 150 ℃; for 5h; Product distribution / selectivity;
|
4 %Chromat. 0.1 %Chromat. |

-

- 1708-29-8
2,5-dihydrofuran

-

- 14631-44-8
3-cyanotetrahydrofuran
| Conditions | Yield |
|---|---|
|
|
14631-44-8 Upstream products
-
19311-38-7

3-chlorotetrahydrofuran
-
143-33-9
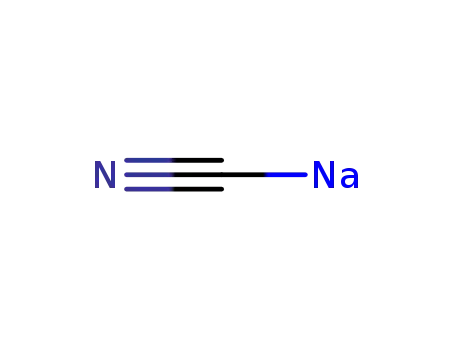
sodium cyanide
-
136421-00-6
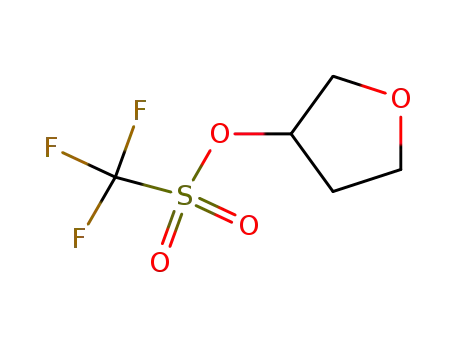
tetrahydrofuran-3-yl trifluoromethanesulfonate
-
13694-84-3
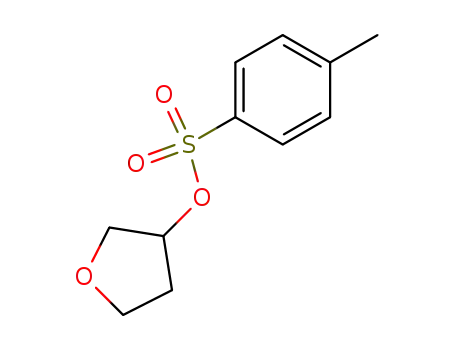
tetrahydro-3-furanyl tosylate
14631-44-8 Downstream products
-
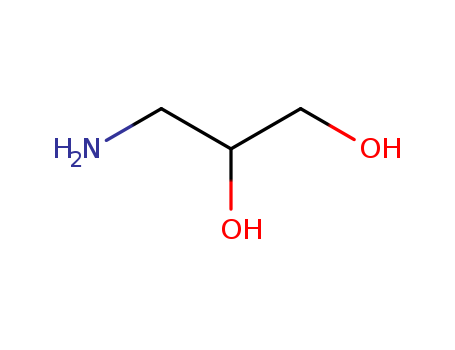
165253-31-6
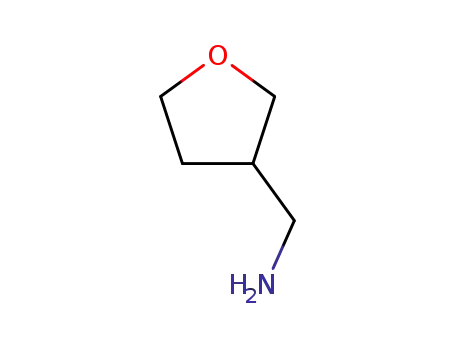
(tetrahydro-3-furanyl)methylamine
-
1092544-69-8
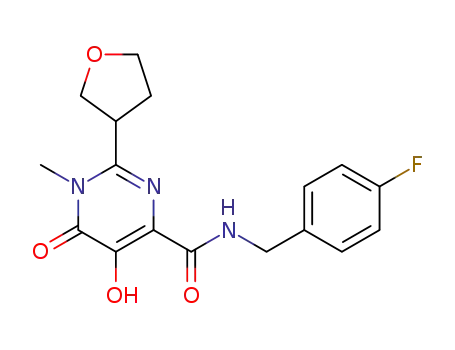
N-(4-Fluorobenzyl)-5-hydroxy-1-methyl-6-oxo-2-(tetrahydrofuran-3-yl)-1,6-dihydropyrimidine-4-carboxamide
-
1092544-81-4
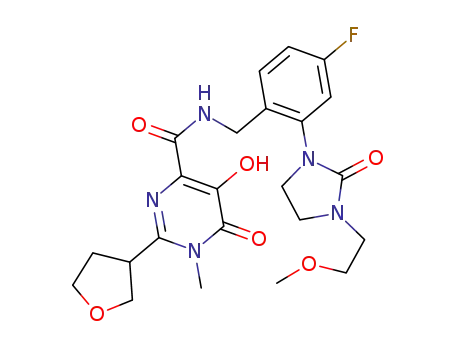
N-(4-fluoro-2-(3-(2-methoxyethyl)-2-oxoimidazolidin-1-yl)benzyl)-5-hydroxy-1-methyl-6-oxo-2-(tetrahydrofuran-3-yl)-1,6-dihydropyrimidine-4-carboxamide
-
1092544-80-3
N-(4-fluoro-2-(3-methyl-2-oxoimidazolidin-1-yl)benzyl)-5-hydroxy-1-methyl-6-oxo-2-(tetrahydrofuran-3-yl)-1,6-dihydropyrimidine-4-carboxamide
Relevant Products
-
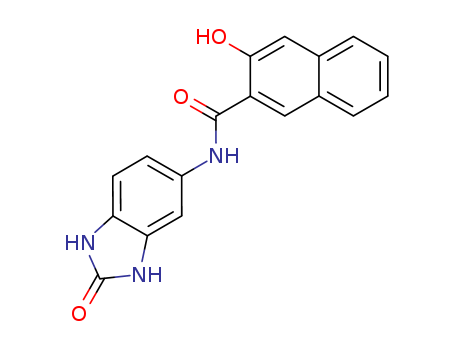
5-(2′-Hydroxy-3′-Naphthamide)-2-Benzimidazolone
CAS:26848-40-8
-
3-Hydroxy-THF
CAS:453-20-3
-
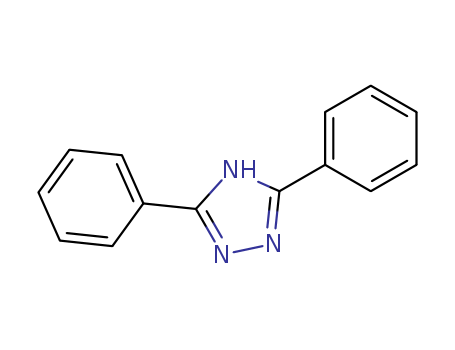
3,5-Diphenyl-1-H-1,2,4-Triazole
CAS:2039-06-7